Lesson 1 – Chemistry
Introduction
Chemistry permeates every aspect of our daily lives, from the myriad of chemical reactions within the human body, powering activities such as digestion and muscle movement, to fundamental processes like photosynthesis in plants, converting sunlight into energy. It extends into the products we use, such as soaps and detergents crafted through chemical processes like sanctification, and even the sunscreens safeguarding us from harmful UV radiation. Chemistry, in essence, is the science that explores the composition, structure, properties, and reactions of matter, providing insights into the interconnections of various scientific disciplines.
I. Definition
1. What is Chemistry?
The study of matter and the components that make it up is the focus of the scientific discipline of chemistry. It also covers these chemicals’ characteristics and the reactions they go through to produce new substances. The main subjects of chemistry are atoms, ions, and molecules, which combine to form elements and compounds. Chemical bonds are typically the means by which these chemical species interact with one another. It is important to note that the interactions between matter and energy are also studied in the field of chemistry.
The study of elements and compounds’ properties, compositions, and structures, as well as how they can change and the energy that is released or absorbed during such changes, is the subject matter of the science known as chemistry. As such, it can be considered a branch of physical science, alongside astronomy, physics and earth sciences including geology.
2. Relationship Between Chemistry and Other Branches of Science:
Chemistry is a branch of science that focuses on the study of matter and its constituent parts. It also discusses the properties of these compounds and the processes by which they react to form new ones. Atoms, ions, and molecules—which come together to form elements and compounds—are the fundamental concepts of chemistry. These chemical species usually interact with each other through chemical bonding.
- The study of language disciplines that are concerned with formal systems is known as the "Formal Sciences." Logic and mathematics are two examples of scientific subjects that fit under this group. regarded as the "language of science."
- The study of natural events through observation and experimentation is the focus of the natural sciences. This area includes biology, physics, and chemistry.
- The study of human communities and the interactions among the individuals who inhabit them is the focus of the social sciences. This group of scientific disciplines includes economics, sociology, and psychology, as examples.
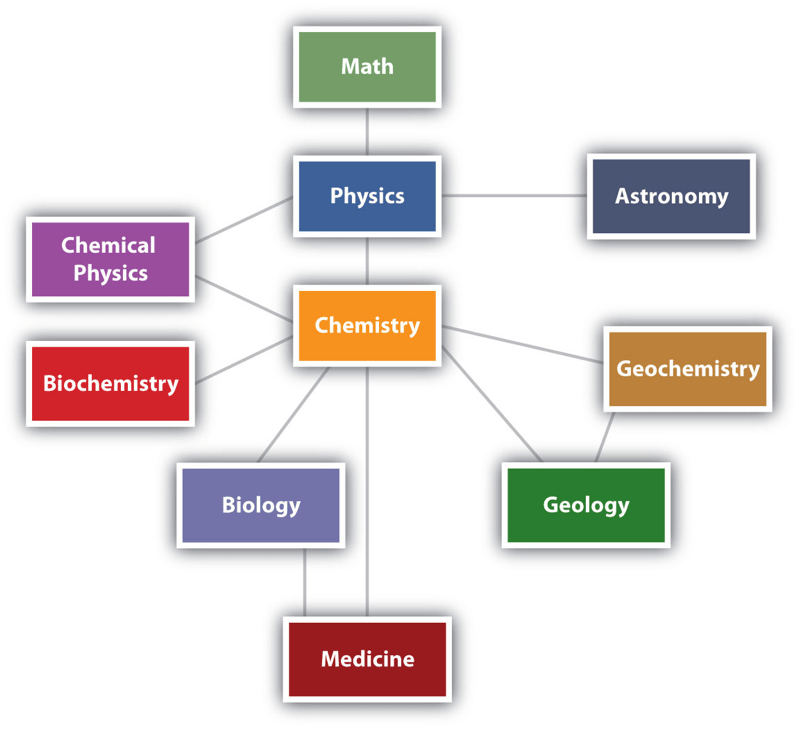
Chemistry is determined to be near the center of the interactions between the major scientific disciplines (as illustrated below). As a result, chemistry can be seen as a foundational science that inspired many other scientific subfields.
II. History of Chemistry
Man has been exposed to changing surroundings ever since he came into existence. He has been quite interested in learning about his surroundings and studying and explaining the things that are happening around him. He has conducted experiments and observations to gather information as a result of his interest. Through the decades, it has also been in charge of many people’s research endeavours around the globe. Systematizing and organising the knowledge acquired in this way was absolutely necessary for the good of humanity. One of the most significant fields of science is chemistry. It turns out reactivity is often largely mediated by the electrons that orbit atoms and the way these are exchanged and shared to create chemical bonds.
1. From Alchemy to Chemistry
A turning point in alchemy was the appearance around the first decade of the fourteenth century of the books attributed to the Arabic alchemist Jabir ibn Hayyan (ca.721– ca.815), known to us as Geber. We find evidence that the speculations of the alchemists were indeed actually based on a large amount of practical knowledge.
However, the practice of chemistry has changed over time along with our understanding of the universe. In other words, for chemistry in its modern form, alchemy is not chemistry. Because Alchemy is a somewhat mysterious and secret method to learn how to control matter. Alchemists used symbols to represent substances, some of which are shown in the accompanying figure. This was not done to better communicate ideas as chemists do today, but to maintain the secrecy of alchemical knowledge, preventing others from sharing that knowledge.
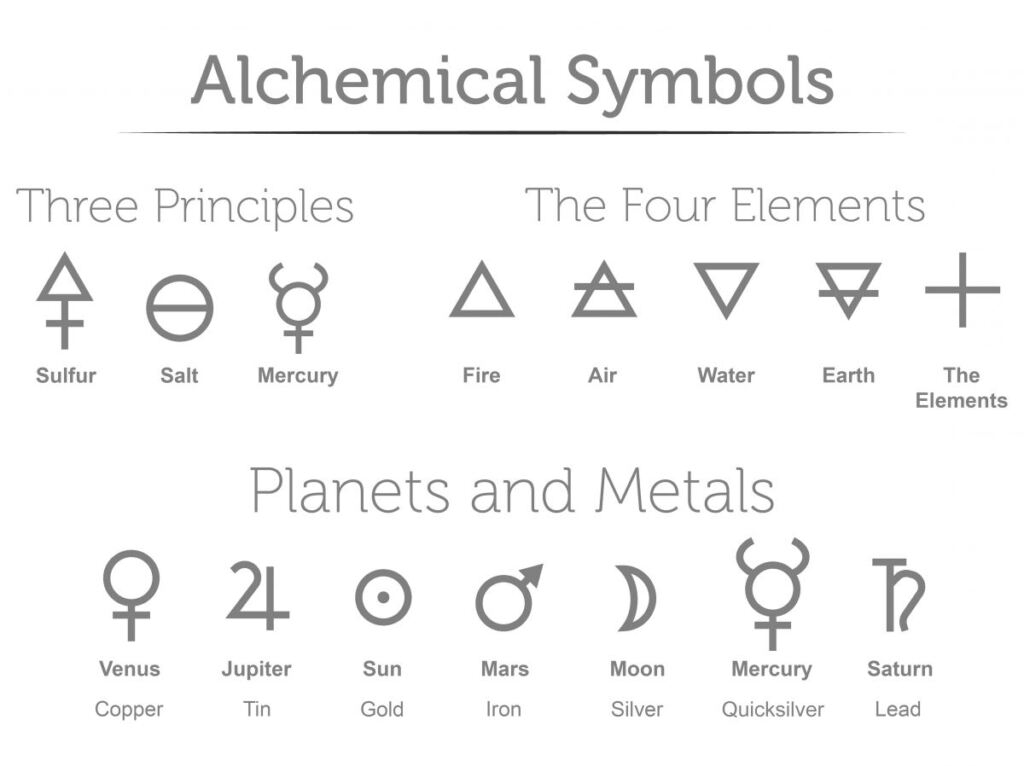
The first affinity table. Table of different relations observed in chemistry between different substances; Memoirs of the Royal Academy of Sciences, p. 202-212. Alchemists used symbols like these to represent substances.
In spite of this secrecy, in its time alchemy was respected as a serious, scholarly endeavor. Isaac Newton, the great mathematician and physicist, was also an alchemist.
2. The Birth of Chemistry As a Science
In this section, we trace the origins of chemistry as a modern science. Without any intention of resuscitating the debate on the defining criteria of science, the term “modern science”––or just “science”––is intended here in its general meaning understood today. Our concept of the meaning of the word “science” was shaped during that crucial period of our history extending over the seventeenth century that is commonly designated with the expression “scientific revolution.” Indeed, the birth of modern science required that our investigations of nature enter a qualitatively different dimension from their pre-scientific antecedents. Even so, the transition was not as sharp as the idea of a “revolution” suggests. There are, in fact, elements of continuity between the scientific investigations of one century and those of the preceding and following centuries. This is particularly true in the case of the development of chemistry, which had to wait until the end of the eighteenth century (after a long process of accumulation of data, technical equipment, and critiques of the old essentialist theories of matter) before acquiring scientific status.
What chemistry acquired with the contribution of Lavoisier was the strict interrelationship between observed facts (experimental data) and theory, which was achieved by astronomy and mechanics a century before with the works of Johannes Kepler (1571–1620), Galileo Galilei (1564–1642), and Isaac Newton (1642–1727). This is the necessary step to close the qualitative gap that divides the pre-scientific from the scientific approach to the investigation of nature. The strict cooperation between observation and theoretical elaboration meant that our reflections on the events of nature abandoned the speculative and qualitative dimension to which they had always been relegated by the various mythological, religious, Aristotelian, and magical explanations of those events, to enter the empirical and quantitative dimension of their prediction and control. This is the conception of science that the scientific revolution brought about.
The anti-scientific nature of magical disciplines like alchemy and astrology, though, should not make us underestimate the important role they played in the laborious process that prepared the way for the scientific revolution. Indeed, their attempts to exercise control over the events of the world, even if carried out in ways that sound bizarre today, had as a consequence an increased attention to the concrete workings of nature. This, in turn, resulted in the growth of the amount of observable facts known and in the development of technical equipment and devices that were later to form the necessary empirical and technological basis for the growth of modern science.
3. The Foundation of Chemical Disciplines
Chemistry is the study of matter and the changes it undergoes and considers both macroscopic and microscopic information. Matter is anything that has mass and occupies space.
It is possible to fix the second period, the birth of chemistry, as a science in the second half of the eighteenth century, with only one exception, the contribution of Robert Boyle (1627–1691), who lived much earlier. The third period, the foundation of chemical disciplines, started around the early nineteenth century with the development of chemistry along the different sectors or disciplines that are still characteristic of present-day chemistry.
4. Modern chemistry
The modern era of chemistry began in the late 19th century when the history of chemistry largely coincided with the history of the contributions of Nobel laureates. This division based on centuries is not official but is evidenced by the fact that in the late 18th and 19th centuries many new revolutionary ideas were developed, radically changing the course of chemical thought.
Chemists help us understand the nature and properties of the world around us, and the history of chemistry is full of discoveries that have fueled this. For example: Antoine Lavoisier paved the way for modern chemistry, helping to introduce field structure by developing an orderly and symbolic language, or Dmitri Mendeleev, the Russian who in 1869 was credited with creating created the periodic table, making it possible to predict how any element will react with another element, and not without mentioning the scientist who provided us with cancer treatments , improving our understanding of radioactive elements and developing portable X-rays for use in field hospitals – and that’s just Marie Curie,…
More recently, advances in chemistry and biology have contributed to the development of vaccines against coronavirus, using our knowledge of DNA and RNA to create the first messenger RNA (mRNA) vaccine approved.
III. Branches of Chemistry
1. Areas of Chemistry
The study of modern chemistry has many branches, but can generally be broken down into five main disciplines, or areas of study: physical chemistry, organic chemistry, inorganic chemistry, analytical chemistry, and biochemistry.
1.1. Organic Chemistry
Organic chemistry, the study of compounds which contain carbon, connects up molecules in new ways to build and analyse an array of materials, from drugs to plastics to flexible electronics.

The main focus of organic chemistry is to isolate, purify, and study the structure of these naturally occurring substances. Examples include formic acid (HCO2H) in ants, ethyl alcohol (C2H5OH) in fermented fruits and oxalic acid (C2H2O4) in rhubarb leaves. Other natural products, such as penicillin, vitamin B12, proteins and nucleic acids, are extremely complex. Organic chemistry is the largest field of specialization among the various fields of chemistry.
1.2. Inorganic Chemistry

Inorganic chemistry is the study of materials primarily based on elements other than carbon. Inorganic compounds can be colorants, fertilizers, catalysts, and more.
Many of the most exciting developments in inorganic chemistry have narrowed the gap with other disciplines. Chemists have also created large inorganic molecules that contain a core of metal atoms, such as platinum, surrounded by a shell of different chemical units. Some of these compounds, called metal clusters, have the properties of metals, while others react in ways similar to biological systems. Small amounts of metals in biological systems are essential for processes such as respiration, neurological function and cell metabolism. Processes of this type are the subject of study in bioinorganic chemistry.
Although organic molecules were once considered a distinctive chemical feature of living organisms, it is now known that inorganic chemistry also plays an important role.
1.3. Physical Chemistry
Physical chemistry involves looking at chemistry through the lens of physics to study changes in pressure, temperatures and rates of conversion, for example, as substances react.

The main branch of physical chemistry devoted to this subject is theoretical chemistry. Theoretical chemists widely use computers to help them solve complex mathematical equations. Other branches of physical chemistry include chemical thermodynamics, which deals with the relationship between heat and other forms of chemical energy, and chemical kinetics, which seeks to measure and understand the rates of reactions chemistry. Electrochemistry studies the interrelationship of electric current and chemical change. There is currently much interest in devices that can use sunlight energy to drive chemical reactions whose products are capable of storing energy. The discovery of such devices would make widespread use of solar energy possible.
A rapidly growing subfield of physical chemistry is surface chemistry. It tests the chemical properties of surfaces, primarily relying on instruments that can provide chemical profiles of those surfaces. Whenever a solid comes into contact with a liquid or gas, an initial reaction occurs on the surface of the solid and as a result its properties can change significantly. Aluminum is a case in point: it is resistant to corrosion precisely because the surface of the pure metal reacts with oxygen to form a layer of aluminum oxide, which protects the interior of the metal from oxidation. further oxidation. Many reaction catalysts perform their function by providing a reactive surface on which substances can react.
1.4. Biochemistry
Biochemistry is the study of the chemical processes that take place in living organisms, for instance in farming, and on the effect the resulting produce will have on our body’s metabolism.

The molecular basis of biologic processes is an essential feature of the fast-growing disciplines of molecular biology and biotechnology. Chemistry has developed methods for rapidly and accurately determining the structure of proteins and DNA. In addition, efficient laboratory methods for the synthesis of genes are being devised. Ultimately, the correction of genetic diseases by the replacement of defective genes with normal ones may become possible.
1.5. Analytical Chemistry
The importance of analytical chemistry has never been greater than it is today. The demand in modern society for a wide variety of safe foods, affordable consumer goods, abundant energy, and labor-saving technology places a heavy burden on the environment. All chemical manufacturing generates waste other than the desired waste, and waste disposal is not always done carefully. Once the amount of a contaminant considered hazardous has been assessed, it is important to detect harmful substances at concentrations well below the hazardous level. Analytical chemists seek to develop increasingly precise and sensitive techniques and instruments.
Sophisticated analytical tools, often combined with computers, have improved the precision with which chemists can identify substances and reduced limits of detection. A commonly used analytical technique is gas chromatography, which separates the different components of a gas mixture by passing the mixture through a long, narrow column containing absorbent but porous material. A gas chromatograph-mass spectrometer can rapidly identify individual components of a chemical mixture whose combined concentration may be no greater than a few parts per billion. Similar or even higher sensitivity can be obtained under favorable conditions using techniques such as atomic absorption, polarization, and neutron activation. The pace of tool innovation is such that analytics tools often become obsolete within 10 years of their introduction. Newer instruments are more accurate, faster, and widely used in the fields of environmental chemistry and pharmaceuticals.

In addition to these major branches, there exist a number of specialized fields of chemistry that deal with interdisciplinary problems. Some such examples include medicinal chemistry, neurochemistry, materials chemistry, nuclear chemistry, environmental chemistry, polymer chemistry, and thermochemistry.
In fact, many civilizations have contributed to the development of chemistry. A lot of early chemistry research focused on practical applications specializing in fields, then known combinations of major disciplines, such as biological organic chemistry or physical organic chemistry. For example, New Materials and batteries are some products of modern chemistry.
2. Examples of Chemistry in Our Daily Lives
We know that all matter is made up of atoms, so chemistry is part of our life because it is present in all the fundamental aspects of our daily life (what we do every day, voluntarily or involuntarily). Chemistry plays a vital role in our daily life. The variety and quality of personal care products, canned foods, computer circuits, television screens, the colors of the houses, the cold of the refrigerator, cleansing agents to clean our house, clothes, etc., are also composed of chemicals. Here are a few more instances of chemistry that occur in people’s daily lives.

- To convert molecules into nutrients the body can absorb and use, digestion depends on the chemical interactions between food, acids, and enzymes.
- Minerals and vitamins are common substances that humans require to survive.
- Chemicals called hormones aid in our development, healing, feeling of self, and search for love.
- Emulsifiers such as soaps and detergents encircle dirt and filth, allowing it to be removed from dishes, clothes, and our bodies through washing.
- Combustion is a chemical reaction that results in fire. It heats houses, cooks meals, and powers cars.
- Chemistry is the reason why drugs work. The chemical compounds may target substances present in infections but not human cells (such as antibiotics) or they may fit into the binding sites for naturally occurring molecules in our bodies (such as blocking pain receptors).
- Food undergoes a chemical transformation during cooking that improves its flavor, eliminates harmful microbes, and increases its digestibility. Cooking heat causes sugars to caramelize, proteins to denature, and other chemical reactions between ingredients.
- Chemicals called cosmetics are applied to our skin to enhance its appearance.
- Certain chemicals are used to manage unwanted plants and animals; they include insecticides, herbicides, and pesticides.
- Petroleum is typically the starting point for chemical processes that produce synthetic textiles and polymers.
- Paint is an intricate concoction of vibrant ingredients.
- A group of compounds called perfume alter our sense of smell.
Conclusion
The impact of applied chemistry research on improving our quality of life is immense, spanning from historic breakthroughs to recent innovations. Throughout history, chemistry has unveiled the mysteries of the world around us, with notable figures like Marie Curie revolutionizing healthcare with portable X-rays and cancer treatments, and Rosalind Franklin’s pivotal role in elucidating the structure of DNA. More recently, the convergence of biology and chemistry has led to the development of mRNA vaccines against coronaviruses, showcasing the power of understanding DNA and RNA. Moreover, chemistry’s influence extends to everyday essentials like plastics, nylon, medications, and even the screens we read from, underscoring its indispensable role in shaping modern living.


